The Bible answers life's spiritual questions and much more. Today, there can be confusion over how to live healthy lives. But we can, and should, turn to God's Word for answers according to two leading health experts.
In Israel, clowning is serious business. The Dream Doctors Project, Medical Clowning in Action, is comprised of a group of more than 100 healing professionals. They are professional members of medical teams at about 30 hospitals in that nation, and focus mostly on juvenile patients. They've been doing it for more than 20 years...
As more states legalize marijuana, new research shows that a greater number of users are struggling with serious health consequences. And many who were hoping for its promised medicinal benefits aren't finding the relief they expected.
Although deadly fentanyl overdoses have decreased recently, they remain the leading cause of death for Americans age 18 to 45. Due to what are known as counterfeit pills, 92 percent of those deaths were accidental. Christian parents are speaking out about the deaths of their beloved children, who were good kids, but made deadly mistakes.
A social media giant has been fined hundreds of million dollars in a landmark case, accused of failing to protect kids online.
A doctor is continuing to sound the alarm on the dangers of marijuana, noting new research is dismantling claims from proponents arguing weed is safe and legality has been a positive move.
In the last two years, the number of college students who identify as transgender has dramatically diminished, according to a report called "The Decline of Trans and Queer Identity among Young Americans."
The debate of gender reassignment procedures for our nation's children is once again front and center. President Trump wants the federal government to take action against these experiments, making it a top issue going into the mid-term elections
Several major medical groups are changing their position on children getting transgender surgery, now advising against it. Surgeon and whistleblower Dr. Eithan Haim told CBN News this could be a watershed moment, saying too many physicians who swore to "do no harm" have broken that oath.
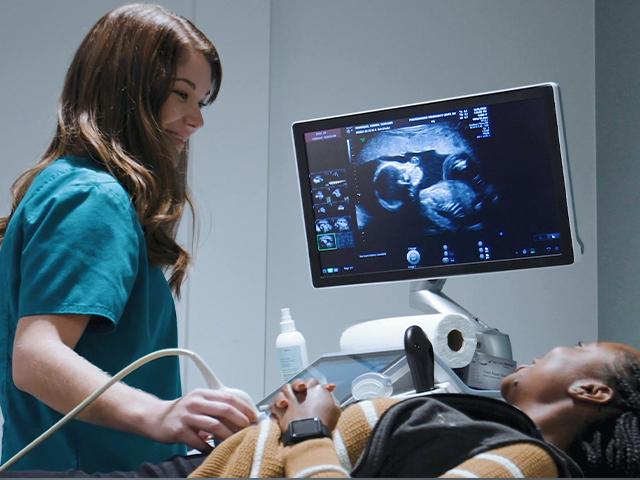
Pro-life pregnancy centers are known for offering ultrasounds, diapers, and other help to women in crisis pregnancies. Recently, a growing number of these centers have expanded their services to include primary medical care.